
With a list of characteristics and applications long enough to make most bioengineers take notice, ultrashort peptide hydrogels are fast becoming the toast of biomedicine. Here, a team from Institute of Bioengineering and Nanotechnology tell us about their work on these little miracles…
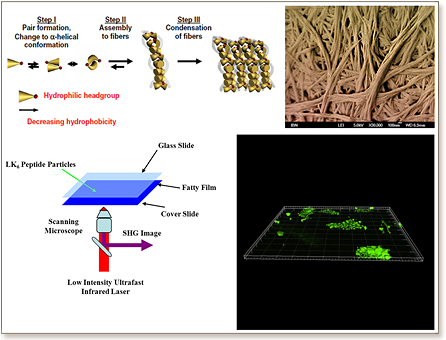
Uncertainties over the long-term effects of artificial materials on the human body has encouraged researchers to work relentlessly in developing natural materials for biomedical applications. Among the vast range of natural building block materials, peptides are highly promising candidates. This can be credited to technologies such as solid- and fluid-phase peptide synthesis, native chemical ligation, as well as synthetic biology technologies, all of which permit the facile synthesis of peptides. Nonetheless, the large-scale economical production of peptide-based biomaterials remains a challenge. The bottleneck lies in the purification stage, whereby desired peptides are isolated from their shorter constituents.
Recent discoveries in our laboratory have unravelled a class of ultrashort peptides that have an innate tendency to self-assemble in water or other aqueous solutions such as phosphate-buffered saline into hydrogels1. Compared to other classes of self-assembling peptides used as biomaterials, these peptides are significantly shorter. Remarkably, hydrogels made from these peptides display scaffold topographies that are similar to those found in collagen-based extracellular matrix (ECM). This structural similarity fuelled our interest in evaluating their potential for biomedical applications, where ECM-like structures are crucial and in demand.

"KAUST shall be a beacon for peace, hope and reconciliation, and shall serve the people of the Kingdom and the world."
King Abdullah bin Abdulaziz Al Saud, 1924 – 2015
Thuwal 23955-6900, Kingdom of Saudi Arabia
Al-Haytham Building (Bldg. 2)
© King Abdullah University of Science and Technology. All rights reserved